
CES2022, the world’s largest technology conference, featured 2,300 companies including a handful of neurotech companies. Of those companies, one attracted a great deal of attention with the demonstration of their proprietary EEG device “iSyncWave.” The company goes by the name of iMediSync.
We interviewed Seung-Wan Kang, CEO of iMediSync, about the company's technology behind the scenes and its future prospects.
About iMediSync
The company provides its own EEG device "iSyncWave" as well as a EEG analysis AI and cloud-based platform.
 iSyncWave
iSyncWave
At CES2022, booth visitors had their brain activity measured and processed into approximately four sheets of diagnostic results in PDF format.
 PDF of diagnostic results obtained from CES2022 demo (report on HRV)
PDF of diagnostic results obtained from CES2022 demo (report on HRV)
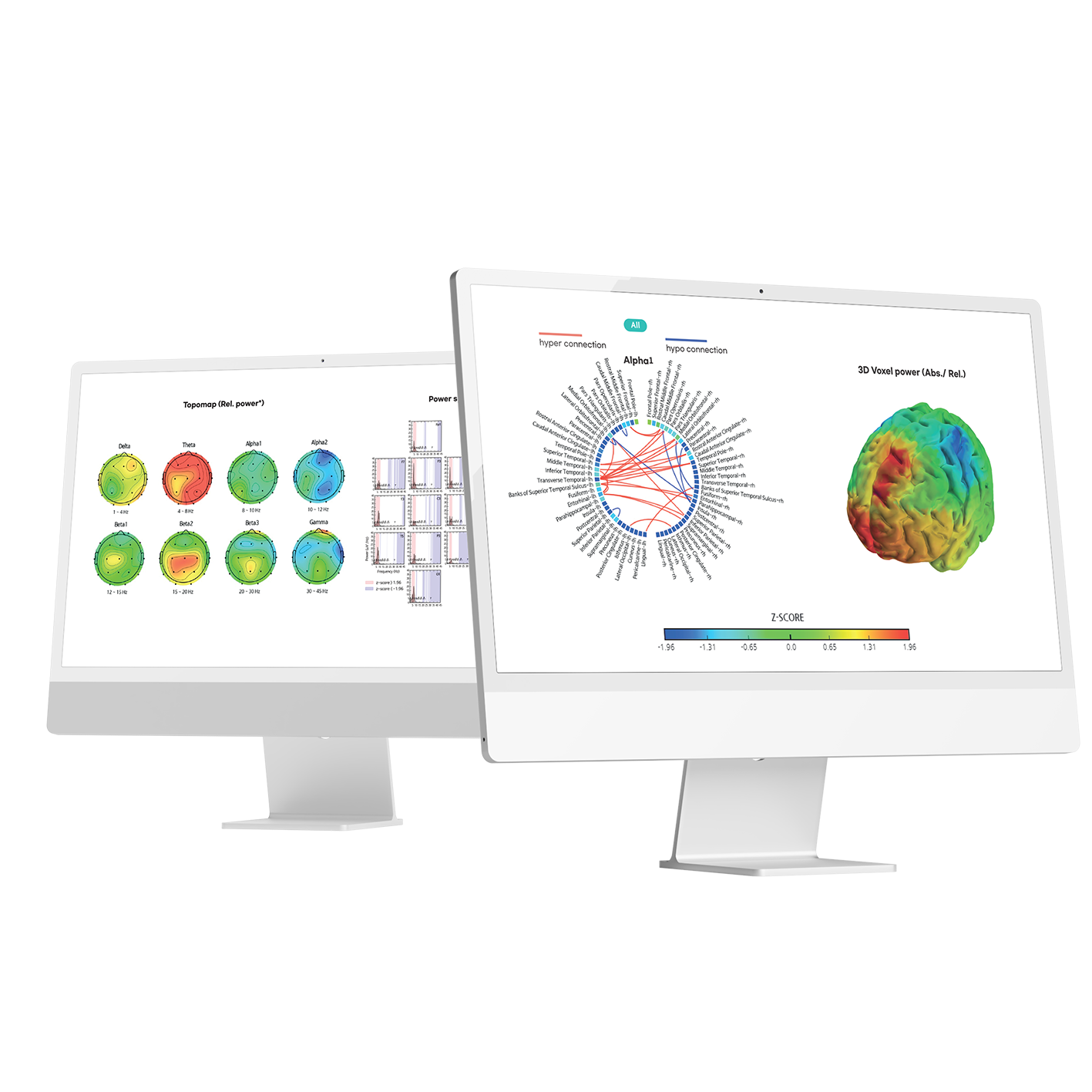 Image: Diagnostic results of EEG
Image: Diagnostic results of EEG
The company was founded in 2012 by Seung, who worked as a physician specializing in pain at Seoul National University Hospital.
The company was founded when he realized that there was a pressing need for an ethical at-home brain imaging solution.
Chronic pain is basically a combination of emotional and cognitive problems. Therefore, it is common practice to diagnose symptoms by measuring brain function. However, ethical issues prevent us from measuring brain function in some patients. I realized that we needed a solution for brain function measurements that could be quantitatively measured on an individual basis for each patient.
The Path to Providing a Service Platform
After deciding to create a functional brain mapping solution, Seung says there have been several steps in the trajectory of the project: creating the EEG database, developing the cloud-based software, and developing the device.
I decided to develop a functional brain mapping solution that crosses EEG with existing technology and data science. The first step was to organize a national project to build a database to store EEG data by gender, followed by the development of a cloud-based EEG analysis platform as a SaaS tool. We then developed an all-in-one device that combines diagnosis and treatment into one.
There are other methods of measuring brain activity, such as MRI, as well as EEG, as a way to map brain function and diagnose brain-related symptoms. Why does the company use EEG?
The changes in emotional and cognitive functions that we focus on are also reflected in EEG. By measuring EEG, we can measure emotional and cognitive states. Additionally, EEG is a good match for neurofeedback using stimulation techniques because we can get data in real time.
The company’s strength lies in not only the EEG measurements, but its ability to perform LED-based stimulation with the same device.
Why did Seung decide to implement brain stimulation into the device?
I have been interested in neurofeedback from the beginning. Stimulation technology using LEDs or electricity can help support the brain's own recovery functions. For example, if you twist your leg and stimulation therapy can reduce the pain as much as possible, then the brain can then control the further treatment and feedback by the brain itself, leading to an better recovery from the pain.
Much research has been conducted on brain-mediated stimulation techniques. Stimulation using electricity or magnetism is considered a common method, and many companies have succeeded in commercializing their products.
Why did the company focus on LED light technology when there are so many different methods of stimulation?
Seung says that the company chose LED technology because of the minimal number of devices required and the ease with which light can be irradiated to the targeted brain region.
Magnetic stimulation (TMS,etc.) is generally difficult to use at home due to its size, and electrical stimulation (tDCS,etc.) is difficult to apply electrical stimulation to targeted areas. However, with light stimulation using LEDs, the device can be made much smaller, and light can be easily directed to a targeted specific location.
Challenges in device design
iSyncWave has won the international product design Red Dot Design Award in 2022.
Although iSyncWave combines many functions in addition to its design, there were many challenges to its device design.
The most difficult part was the device design. We wanted to make sure that the electrodes were placed in close contact with the correct positions on the scalp. We needed the device to automatically fit along the shape of each individual's head.
Hayato, the author of the article, actually wore the device at CES2022 and felt the device automatically fitting to the shape of his head. Therefore, the brain waves displayed in real time contained relatively low noise.
 At the booth on CES2022, measuring brain waves and other biometrics data
At the booth on CES2022, measuring brain waves and other biometrics data
Looking to expand overseas
CES attracts visitors from all over the world in a variety of industry domains. Therefore, companies that exhibit at the event may be able to attract clients from outside their home countries.
iMediSync is no exception. Thanks in part to the attention-grabbing demonstration, the company has received business offers for joint development and service purchases from many companies in Europe, the United States, Australia, and other countries, not limited to Asian countries. They are planning to expand overseas in the future.
The company also received the approval of pre-market clearance (510k) from the FDA last month to sell its medical devices in the US. As a result, the company's devices will soon be distributed in the United States. The company is also planning to apply for a medical device cetification in Japan.
Conclusion
In this article, we interviewed Seung-Wan Kang, CEO of iMediSync, about the company's path to developing the services it offers.
The company is on the right track, but they faced their fair share of tough decisions and challenges to get there. This is why the company is now able to offer their innovative devices and platforms.
The company's future development, as it looks to expand overseas, will also be interesting to watch.


